Health effects of formaldehyde in Europe
| This page is a product of the EBoDE project. The final report of the EBoDE project has been published as a report in 2011[1] and also as web pages in Opasnet. These links lead to parts of the report.
EBoDE project: main page | overview | contributors | data overview | Parma meeting | abbreviations | all pages Methods: environmental burden of disease calculation | selection of exposures and health effects | data needed | impact calculation tool Health effects in Europe: benzene | dioxins | formaldehyde | lead | ozone | particulate matter | radon | second-hand smoke | transport noise | environmental burden of disease | results by country |
This page is a study.
The page identifier is Op_en5205 |
|---|
| Moderator:Pauli (see all) |
|
|
| Upload data
|
About formaldehyde
Formaldehyde is a high-production volume chemical widely used in building materials, industrial processes and wide range of products. Formaldehyde is widely present both indoors and outdoors, but it reaches high levels mostly indoors. It is used in the production of several building materials and household products, or it can be a by-product of combustion. The high volatility of the compound can lead to high formaldehyde levels in indoor spaces.
Predominant acute symptoms of formaldehyde exposure in humans are irritation of the eyes, nose and throat and aggravation of asthma symptoms (WHO, 2000a). A number of studies point to formaldehyde as an important indoor irritant associated with respiratory illness. A relationship between asthma-like symptoms and indoor concentrations of formaldehyde has been reported, as well as between exposure to formaldehyde emitted from indoor paint and asthma. Repeated exposures are not associated with more severe effects or lowering of the threshold concentration. Consequently, short-term concentrations are predictive of the effects also after long-term exposure.
Exposure to formaldehyde has also been associated with development of cancer. Convincing evidence exists of high concentrations of formaldehyde being capable of inducing nasal cancer in rats and possibly in mice and genotoxic effects in a variety of in vitro and in vivo systems. Sinonasal cancer in humans has also been associated with high formaldehyde exposures in occupational industrial settings (ranging from 2 to 6 mg m-3) (WHO, 2000a). Based on this, IARC has recently classified formaldehyde as carcinogen group 1 (IARC, 2006a).
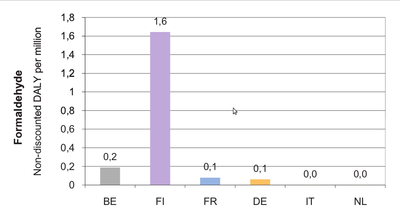
Formaldehyde was included in EBoDE due to its high toxicological potential, economic significance and related political concern. [1]
Selected health endpoints and exposure-response functions
In the EBoDE study, only the development of asthma in toddlers has been included. Sinonasal cancer was not included, because the WHO Air Quality Guidelines working group (WHO, 2000a) as well as recent update of the reviews for the development of WHO Guidelines for indoor air quality (WHO, 2010b) concluded that there is no epidemiological or toxicological evidence that formaldehyde would be associated with sinonasal cancer at levels below 1 mg/m3. The WHO Guidelines for Indoor Air Quality use eye irritation as the main health end-point associated with formaldehyde; however, due to difficulties in estimating a burden of disease from irritation this endpoint was not included in our calculations.
Association with asthma is suggested by the systematic review by McGwin et al., 2010, even though evidence has not been consistent across all the studies (e.g. Krzyzanowski et al, 1990). We selected childhood asthma as the endpoint for formaldehyde, but due to the inconsistencies in the scientific evidence the estimates calculated here should be considered preliminary and to be confirmed by future research. In order to estimate formaldehyde-related asthma, we used the exposure-response function as reported by Rumchev et al. (2002). They studied a cohort of 88 children in Perth, Australia. For every 10 μg m-3 increase in formaldehyde exposure in bedrooms, they found an increase of 3% in the risk of having asthma (OR=1.03, 95% CI 1.02–1.04). Based on a reanalysis of their data over reported exposure categories and rescaling for 1 μg m-3, the relative risk used in our calculation is 1.0167 (see also Table 3-19 in section 3.12). Asthma effects were calculated for children (<3 years). A similar association may potentially exist for older children and adults, but due to the lack of evidence such relationship was not modelled. This may lead to underestimation of the true formaldehyde-related burden of disease.
A threshold level for effects was applied. The original study by Rumchev reported elevated risks starting from exposures of 60 μg m-3. When their data were plotted in order to derive the relative risk, the threshold could be even as low as 40 μg m-3. However, the Rumchev study was criticized for confounding factors. WHO (2000a, 2010b) indicated that the lowest concentration that has been associated with nose and throat irritation in exposed workers after short-term exposure is 0.1 mg m-3, although some individuals can sense the presence of formaldehyde at lower concentrations. To prevent significant sensory irritation in the general population, an air quality guideline value of 0.1 mg m-3 as a 30-minute average was recommended as the WHO Guideline (WHO, 2010b). This is the threshold value that we used in our calculations. Since this is an order of magnitude lower than the presumed threshold for cytotoxic damage to the nasal mucosa, there is a negligible risk of upper respiratory tract cancer in humans below this threshold. As part of the uncertainty analysis, we compared alternative threshold models for cancer (threshold levels of 40, 60 and 100 μg m-3) and asthma, see section 5.2. [1]
Exposure data
Inhalation is the dominant pathway for formaldehyde exposure in humans. The relevant exposure metric is the residential indoor air level (μg/m-3). As indicated above, both of the exposure-response models used apply a threshold level (100 μg m-3). Therefore, it is necessary to assess the fraction of the population being exposed to levels higher than this threshold level. A probabilistic simulation model was used to calculate the fraction of the population exceeding the threshold using mean and standard deviation data and assuming lognormal distributions. No international exposure data sources were identified for formaldehyde, so data have been collected from heterogeneous national sources.
For Belgium, Germany and the Netherlands, only mean exposures were available, without information about the variability. For these three countries, the exposure distributions were based on the data from the other countries (estimated coefficient of variation: 0.6).
| Country | mean
μg m-3 |
sd
μg m-3 |
References |
|---|---|---|---|
| Belgium | 24.0 | 14.41 | Swaans et al,. 2008 |
| Finland | 41.6 | 22.4 | Jurvelin et al, 2001 |
| France | 23.0 | 14.0 | OQAI, 2006 |
| Germany | 26.0 | 15.61 | Umweltbundesamt, 2008 |
| Italy | 16.0 | 8.0 | Lovreglio et al, 2009 |
| Netherlands | 13.0 | 7.81 | Dongen,van & Vos, 2008 |
1 Mean coefficient of variation of the countries with data on variability used for estimation.
Exposure data for formaldehyde are presented in Table 3-21 in section 3.12.
The mean formaldehyde indoor concentrations vary from 13 μg m-3 in the Netherlands to about 42 μg m-3 in Finland. In Finland formaldehyde exposure levels are higher than in many other developed countries due to the construction materials used and the relatively tightly sealed building envelopes. As shown in Figure 3-3, approximately 42% of population is exposed to levels above 40 μg m-3 and 2 % above 100 μg m-3.
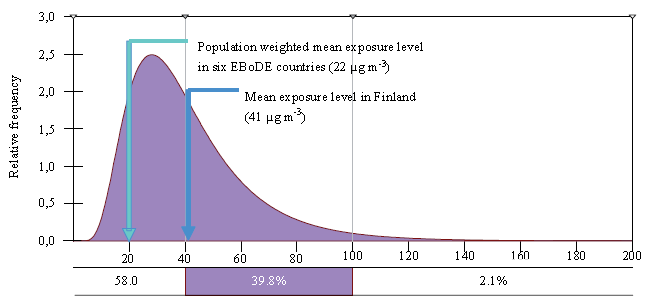
FIGURE 3-3. Estimated formaldehyde exposure distribution in Finland.
Data comparability is compromised for formaldehyde by the differences in population sampling. In France, Germany and the Netherlands, data measurements are representative for country-wide exposure. However, in other countries, measurements have only been carried out in a few cities or were based on a smaller subset of houses. [1]
Uncertainties per stressor and comparison with other studies
A list of the most important sources of uncertainty for each stressor in the EBoDE calculations is provided in Table 5-1. Some of these are further explained below. In addition, we will compare our estimates to results of a selection of similar studies. Comparison of different studies on environmental burden of disease helps to understand the role of various methodological and strategic selections made in each study, like the selection of stressors or health endpoints.
Formaldehyde
No international burden of disease study utilizing DALYs for formaldehyde was identified. WHO Guidelines for Indoor Air Quality used eye irritation as the main health end-point in setting a safe exposure level. However eye irritation cannot be directly used as a health end-point in burden of disease calculation because no disability weight exists and therefore was not accounted for here. Scientific evidence on the association between formaldehyde and childhood asthma is not considered sufficiently consistent yet; thus the results presented here must be taken as provisional estimates of the magnitude of the health impacts, to be confirmed by future studies. [1]
| Excluded health endpoints and related assumptions | Exposure data | Exposure response function | Calculation method | Level of overall uncertainty a) | Likely over- or underestimation b) | |
| Formaldehyde | Acute symptoms; nasopharyngeal and sinonasal cancers. | Data from different years. Population representativity varies. For some countries limited national coverage. Limitation in technique to detect peak exposures | Shape of ERF. Threshold level. Partly inconclusive evidence for the endpoint. ERF from <3 yr olds; potential effects at older ages not accounted for | Simulation of threshold exceedances. Selection of age groups | *** | Underestimation, mainly due to exclusion of = 3year olds but also not accounting for eye irritation |
References
- ↑ 1.0 1.1 1.2 1.3 1.4 Otto Hänninen, Anne Knol: European Perspectives on Environmental Burden of Disease: Esimates for Nine Stressors in Six European Countries,
Authors and National Institute for Health and Welfare (THL), Report 1/2011 [1] [2] Cite error: Invalid
<ref>tag; name "EBoDe" defined multiple times with different content