Health effects of dioxins in Europe
| This page is a product of the EBoDE project. The final report of the EBoDE project has been published as a report in 2011[1] and also as web pages in Opasnet. These links lead to parts of the report.
EBoDE project: main page | overview | contributors | data overview | Parma meeting | abbreviations | all pages Methods: environmental burden of disease calculation | selection of exposures and health effects | data needed | impact calculation tool Health effects in Europe: benzene | dioxins | formaldehyde | lead | ozone | particulate matter | radon | second-hand smoke | transport noise | environmental burden of disease | results by country |
This page is a study.
The page identifier is Op_en5203 |
|---|
| Moderator:Pauli (see all) |
| This page is a stub. You may improve it into a full page. |
| Upload data
|
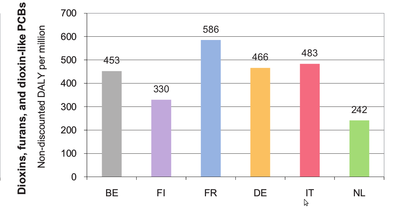
Dioxins (including furans and dioxin-like PCBs) are a group of polychlorinated organic compounds with the same toxic mechanism. They are by-products of various industrial processes and combustion activities and are considered to be highly toxic.
Dioxins and dioxin-like PCBs are quantified by toxic equivalents (TEQs) representing the total toxicity compared to the most toxic compound, 2,3,7,8-Tetrachlorodibenzodioxin (TCDD). The power of toxicity is calculated with Toxic Equivalent Factors (TEFs), which allow the toxic potentials of each compound to be added up, in order to derive the TEQ of the mixture. Acute toxicity, leading for example to chlorakne or alteration of liver function, is only expected at very high doses. Long-term exposure to dioxins has been linked to effects on the immune system, the nervous system, the endocrine system and reproductive functions and is also known to cause tooth and bone defects, diabetes as well as several types of cancer (USEPA, 2003). The association between dioxins and cancer has been most consistent for non-Hodgkin’s lymphoma. IARC classified TCDD (2,3,7,8-Tetrachlorodibenzo-p-dioxin), as a “known human carcinogen” (IARC, 1997). All other dioxin-like compounds are classified as “likely to be carcinogenic to humans”.
This group of chemicals is selected in EBoDE because of their high toxicity and potential troubling exposures through e.g. mothers milk. [1]
Selected health endpoints and exposure-response functions
In EBoDE, we have quantified the effect of exposure to dioxins and dioxin-like PCBs on cancer (all cancer types, mortality only). The non-fatal and non-cancer effects were not suited for health impact assessments due to difficulties in estimating the exposure-response relationships and the other input parameters necessary for estimating DALYs. Therefore, our estimates may underestimate the true dioxin-related burden of disease.
Leino et al. (2008) assumed a linear exposure-response relationship for excess cancers associated with dioxin intake. They estimated the health risk for toxicity equivalent intake assuming additivity of the toxicity of the different types of dioxins and all cancer cases to be lethal.
The EBoDE calculations use the Leino et al. (2008) approach, but the results have been corrected with an updated cancer slope factor 1×10-3 per pg/kg/d of dioxin intake of the U.S. Environmental Protection Agency (USEPA, 2003; NAS, 2006). The assumption that all cancers are lethal may lead to overestimation of the impacts.
The health endpoints considered in this project for dioxins and the corresponding exposure-response functions are summarized in Table 3-19 in section 3.12. YLD estimates in the table are based on the attributable fraction derived from the ERF using method 2A (see Figure 2-1), which is applied to the total YLD for all cancers as represented in the WHO database. [1]
Exposure data
Dioxins and dioxin-like PCBs are persistent and bio-accumulating. The main exposure route for these chemicals is animal fat in nutrition, which accounts for about 90% of all exposure. Other routes, such as inhalation, play a minor role. In order to estimate health effects related to dioxin exposure, daily intake data were needed. This intake depends on eating habits, age, gender, body weight and food consumption. Often, breast feeding contributes to the highest intake of dioxins for humans in their life. Dioxins have a long half life. Therefore the development of health effects in humans depends not only on the daily intake, but also on the body burden accumulated over years. On average, the daily intake of dioxins and dioxin-like PCBs decreases, while the body burden increases with age.
The cancer slope factor is expressed for daily intake of adults. There are different ways to measure the daily intake, each with different limitations. Table 3-2 describes some different measurement methods and provides short information about their use and limitations.
| Type of measurement | Type of use | Specific limitations and uncertainties | |
|---|---|---|---|
| A | Survey (questionnaire) on food consumption | Information on food consumption and about the content of dioxins in representative food samples allow modelling of daily intake | Results are modelled for an average population - food contamination and eating habits can differ on a large scale |
| B | Total diet studies | The total diet in a population group over a certain time period and dioxin in this food or representative food samples are measured. | Results are only relevant for the investigated groups and not necessarily representative for the whole population, sampling period influence the results. |
| C | Human biomonitoring Investigation of human milk or blood levels | Analyses of samples can show the body burden. Experimental scaling is used to convert observed biomonitoring results (blood) into daily intakes. | D-R function is based on daily intake. Human milk or blood samples are not widely available. Different fat content of the bodies influences the results. |
In addition, in all these studies different compounds can be measured:
- Only dioxins and furans;
- dioxins, furans; and dioxin-like PCBs
- dioxins, furans and dioxin-like PCBs as well as all other dioxin-like compounds detected as dioxin-like activity, expressed as TEQ in Bioassays (e.g. CALLUX).
In the EBoDE project, we have used national exposure data because there is no international comparable data source available. The different countries have used different methods to derive the daily intake values.
Table 3-3 provides a summary of the data and sources for dioxin. The specific data used in this project are summarized in Table 3-21 in section 3.12. For the EBoDE project daily intake data are expressed as Toxic Equivalent (TEQ), estimated using the Toxic Equivalent Factors (TEFs) as provided by WHO (Van den Berg et al. 1998). Even though later TEFs exist (Van den Berg et al., 2006; http://www.who.int/ipcs/assessment/tef_update/en/), we used the results of the 1998 review, because most available data have been calculated using these TEFs.
| Countries | Population groups | Source | Sampling years | Compounds measured | Dioxin intake 2004 pg/kg bw/d |
|---|---|---|---|---|---|
| Belgium (A) | female 18-44 y
adults 50-65 y adults |
Bilau 2008
Bilau 2008 Calculated mean |
2002–2006 | Calux-all dioxin-like compounds1 | 2.1
1.7 1.9 (mean) |
| Finland (A) | all | Kiviranta et al 2005 | 2002 | Dioxins+PCB | 1.5 |
| France (C) | 30–65 y | Fréry et al. 2006 | 2004 | Dioxins+PCB | 2.32 |
| Germany (A) | adults | Umweltbundesamt 2005 | 2003 | Dioxins+PCB | 2.0 |
| Italy (A) | 13–94 y | Fattore et al 2006 | 1997–2003 | Dioxins+PCB | 2.33 |
| Netherlands (A+B) | adults | De Mul 2008 | 2004 | Dioxins+PCB | 1.04 |
1 Belgium – Dioxin and all dioxin-like compounds are measured with Bioassay, only the sum of all dioxin-like compounds is given; the daily intake was calculated as mean of the 2 adult groups.
2 France – daily intake calculated based on blood concentration of 27.7 WHO-TEQ pg/g blood fat.
3 Italy – daily intake were calculated using, for most dioxin and DL-PCB concentration data, a database available from the European Commission (Gallani et al., 2004).
4 Netherlands – Values in the study were calculated using TEFs from 2005. For comparability, we have adjusted the values as presented by Mul et al (2008) by adapting the results to TEF 1998 adding 10%.
We have only used data on the daily intake of adults. We have chosen to do so, because the daily intake differs substantially between different age groups. The highest intakes are calculated for breastfed babies (about 50 to 100 WHO-TEQ pg/kg bw/d). Children have a higher intake than adults because of the different proportion between body weight and food intake and their different food habits (children take more milk and dairy products). Since there are only very few data for children available, we have limited ourselves to adults.
Due to the differences in measurement approach, it is difficult to compare dioxin intake numbers between countries. As a form of quality assurance, we have compared our daily intake estimates of dioxins and dioxin-like PCBs to international data on dioxins and PCBs in mother's milk (milk data from 2001–2003) as provided by WHO in the ENHIS-database (WHO, 2007a) and from Malisch and Leeuwen (2003). In principle, the ratio between the estimated daily intakes and the levels of mother’s milk should be roughly similar between countries. The ratios are presented in Table 3-4. As can be seen from this table, the ratios are relatively similar across the countries, except in the Netherlands, where the intake level seems to be somewhat lower than in the other countries in comparison with the mother’s milk levels. We have not corrected for this difference in the EBoDE calculations, as the causes for the difference are yet unknown. [1]
| Countrya | Human milk
ng TEQ/kg fat |
Daily intake
pg TEQ/kg bw/d |
Factor
milk/intake |
|---|---|---|---|
| Belgium | 29.5 | 1.9 | 16 |
| Finland | 15.3 | 1.5 | 10 |
| Germany | 26.2 | 2.0 | 13 |
| Italy | 29.0 | 2.3 | 13 |
| Netherlands | 29.8 | 1.0 | 30 |
a France was not included in the WHO-milk study.
Uncertainties per stressor and comparison with other studies
A list of the most important sources of uncertainty for each stressor in the EBoDE calculations is provided in Table 5-1. Some of these are further explained below. In addition, we will compare our estimates to results of a selection of similar studies. Comparison of different studies on environmental burden of disease helps to understand the role of various methodological and strategic selections made in each study, like the selection of stressors or health endpoints.
Dioxins Our calculations were based on the same approach as applied earlier by Leino et al (2008), but we utilized an updated cancer slope factor that is approximately seven times higher than the one used by Leino et al. Leino et al. did the calculations for Finland only. The work presented here also updated the exposure estimates in order to allow for good international comparability, yet some differences between the national intake estimation methods remained.[1]
| Excluded health endpoints and related assumptions | Exposure data | Exposure response function | Calculation method | Level of overall uncertainty a) | Likely over- or underestimation b) | |
| Dioxins (plus furans and PCBs) | Effects on the immune, endocrine, reproductive and nervous system; tooth and bone defects. All cases of cancer assumed to be fatal. | Indirect exposure metrics. Different measurement methods. Daily intake of food depends on age, body weight and eating habits. Exposure varies within countries (from region to region) | Uncertain cancer slope factor. Assumed additivity of the toxicity of different types | UR method of calculating PAF results in overestimation because all cases are assumed to be fatal. | *** | Underestimation of non cancer effects, Overestimation of cancer effects (all lethal) |
References
- ↑ 1.0 1.1 1.2 1.3 1.4 Otto Hänninen, Anne Knol: European Perspectives on Environmental Burden of Disease: Esimates for Nine Stressors in Six European Countries,
Authors and National Institute for Health and Welfare (THL), Report 1/2011 [1] [2] Cite error: Invalid
<ref>tag; name "EBoDe" defined multiple times with different content