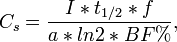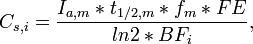Infant's indirect exposure
| Moderator:Jouni (see all) |
|
|
| Upload data
|
Contents
Question
How to measure infant's dioxin exposure due to fetal period and breast feeding?
Answer
Mother's exposure at level 100 pg /d (fairly typical amount) will lead to child's TEQ concentration of 44 pg /g fat (logTEQ 3.8).
Rationale
- The equations and code below was developed for dioxins, but later it was extended to perfluorinated alkyl substances (PFAS) as well.
Transformation between serum concentration and intake:
where Cs = serum concentration of compound in pg/g fat I = average daily intake of compound in pg/kg/day t1/2 = the half-life of compound (2737.5 d = 7.5 a) f = fraction of ingested compound actually absorbing from the gut (0.80) BF% = body fat percentage a percentage of total daily dietary intake of compounds that come from fish (0.86).
The previous equation applies in a single individual. In the case of dental aberrations, the main exposure comes from the mother during pregnancy and breast feeding. For this, we use
where Cs,i = serum concentration of compound in the infant in pg/g fat Ia,m = average daily intake of compound of the mother in absolute amounts pg/day t1/2,m = the half-life of compound in the mother (2737.5 d = 7.5 a) fm = fraction of ingested compound actually absorbing from the gut in the mother (0.80) FE = fraction of mother's compound load that is transported to the infant during breast feeding (0.25) Vartiainen et al. REF BF = body fat amount in the infant (into which the compound is evenly distributed) during the period when tooth are sensitive to defects and the exposure at its highest (ca. six months of age) (1 kg)
With dioxin, body fat represents the distribution volume, as the compound distributes evenly in the fat compartments of the body. Therefore, the dioxin concentration per gram fat is approximately the same in blood and fat tissue. With PFAS, the concentration in blood equals total amount divided by the distribution volume.
Inputs
| Obs | Exposure_agent | Parameter | Value | Description |
|---|---|---|---|---|
| 1 | TEQ | half-life | 2737.5 | t0.5: 7.5 * 365, Dioxin half-life in adults (d) |
| 2 | TEQ | fraction absorbed | 0.6 - 0.8 | f_ing: 0.70, Fraction of compound ingested that is actually absorbed (-). Adjusted downwards to reflect KTL sarcoma study concentrations |
| 3 | TEQ | fraction to child | 0.05 - 0.1 | f_mtoc: 0.075, Fraction of mother's compound burden that enters the child during breast-feeding in 6 mo. Alaluusua et Vartiainen used 0.25 but that produces way too high predictions (-) |
| 4 | TEQ | distribution volume in infant | 5400 | BF: 0.2 * 26.923 kg, body fat amount in 9-year-old boys |
| 5 | PFAS | half-life | 1642.5 (912.5 - 3285) | t0.5: 4.5 (2.5 - 9) a * 365, PFOS half-life in adults (d), EFSA, slightly adjusted |
| 6 | PFAS | fraction absorbed | 0.1 - 0.15 | f_ing: 0.80, Fraction of compound ingested that is actually absorbed (-) See below. |
| 7 | PFAS | fraction to child | 0.2 - 0.4 | f_mtoc: 0.25, Fraction of mother's compound burden that enters the child during breast-feeding in 6 mo. Alaluusua et Vartiainen (-) |
| 8 | PFAS | distribution volume in infant | 1580 | BF: 200 ml/kg for PFOS * (7.692 kg + 8.141 kg) / 2, body fat amount in the infant at six months of age (g) [[:op_en:Body weight of 0-24 months old Finnish children]]. |
| 9 | MeHg | half-life | 49.5 | t0.5: elimination from blood: t0.5 = ln 2 / elimination rate = ln 2 / 0.014 /day = 49.5 d |
| 10 | MeHg | fraction absorbed | 0.95 | f_ing: 95 % absorbed |
| 11 | MeHg | fraction to child | 0.25 | f_mtoc: the hair-to-blood Hg concentration ratio (0.25 L blood/g hair) |
| 12 | MeHg | distribution volume in infant | 84.7 | BF: 5 l / 0.059 = 84.7 l |
| 13 | DHA | half-life | 1 | t0.5: nominal bacause the ERF is defined as mother's intake rather than child's concentration |
| 14 | DHA | fraction absorbed | 1 | f_ing: nominal |
| 15 | DHA | fraction to child | 1 | f_mtoc: nominal |
| 16 | DHA | distribution volume in infant | 1 | BF: nominal |
Dioxin kinetics
Previously we looked at the infant at six months of age, because that was arguably the age with peak dioxin concentration. The amount of breast milk consumed is likely to decrease and also the body dilution reduces concentrations. This would be a relevant time point for the dental aberration effect. However, EFSA considers the sperm concentration as the most sensitive endpoint of dioxins, and that ERF is based on dioxin concentration in 9-year-old boys. Therefore, we try to estimate dioxin concentration at the age of 9 years to be comparable to the EFSA ERFs.
Body weight data comes from Body weight of 0-24 months old Finnish children and Body weight of 2-18 year old Finnish children.
Direct exposure (TEQ pg/d to mother) is converted to infant's TEQ concentration (pg/g fat).
PFAS kinetics
According to EFSA[1], a long-term exposure of 4.4 ng/kg/d to PFAS leads to 6.9 ng/ml in blood. This implies that fm = 6.9 ng/ml / 4.4 ng/kg/d / 1642 d * 200 ml/kg * ln2 = 13 % of PFAS is actually absorbed by the mother.
This in turn leads to PFAS concentration of 17.5 ng/ml blood in the child, i.e. ca 2.5-fold compared with the mother. This implies that FE = cs,i / amount in mother * BFi = 17.5 ng/ml / (6.9 ng/ml * 200 ml/kg * 70 kg) * (200 ml/kg * 8 kg) = 0.29.
Direct exposure (PFAS ng/d in mother) is converted to infant's PFAS concentration in blood (ng/ml).
Methylmercury kinetics
See ERF of methylmercury for EPA kinetics[2]. The conversion from mother's intake to mother's hair concentration is I' * 0.200 d/g, where I' is the daily intake (total, NOT per kg bw).
Direct exposure (MeHg ug/d in mother) is converted to mother's MeHg concentration in hair (ug/g).
Calculations
- Ovariables initiated 19.5.2017 [1]
- Updated 30.5.2017 [2]
- This code was called initiate with dx.expo.child until 13.6.2019. Now it has expo_indir for indirect exposure estimates. Indirect here means that the exposure is not directly estimated from environmental concentrations but indirectly from mother's exposure.
- An updated code expo_indir2 includes PFAS as well as dioxin. 2021-01-20 [3]
See also
- Human PBPK model for dioxin
- ↑ EFSA. (2020) Risk to human health related to the presence of perfluoroalkyl substances in food. https://doi.org/10.2903/j.efsa.2020.6223
- ↑ Integrated Risk Information System (IRIS). Chemical Assessment Summary, 2001. [4] [5] Accessed 13.9.2019